Natural penicillins
Objectives:
Describe the basic chemical structure of penicillins
Explain how the side chain determines spectrum and stability
Describe the mechanism of action involving PBPs
Explain peptidoglycan synthesis and cross-linking
List and explain the four mechanisms of β-lactam resistance
Classify β-lactamases by Ambler molecular class
Differentiate the five classes of penicillins by spectrum
Describe pharmacokinetic properties and dosing adjustments
Recognize adverse effects and hypersensitivity reactions
Select appropriate β-lactam/β-lactamase inhibitor combinations
Link to recorded lecture
Quick Reference: Penicillin Dosing
Penicillin G
- Usual Adult Dose: 8–24 Million Units/Day IV in equally divided doses every 4–6 hours
- Renal/hepatic failure: decrease dose in renal failure
- CSF penetration: poor (5–10% with inflamed meninges)
- Adverse effects: hypersensitivity, hyperkalemia (K⁺ salt), hypokalemia (Na⁺ salt)
Penicillin V
- Usual Adult Dose: 250–500 mg every 6–12 hours orally
- Renal/hepatic failure: decrease dose in renal failure
- CSF penetration: poor
- Adverse effects: hypersensitivity, nausea/vomiting
Oxacillin
- Usual Adult Dose: 2 g every 4 hours IV
- Renal/hepatic failure: no adjustment needed
- CSF penetration: poor
- Adverse effects: hypersensitivity, hepatotoxicity, interstitial nephritis
Nafcillin
- Usual Adult Dose: 2 g every 4 hours IV
- Renal/hepatic failure: no adjustment needed (hepatic elimination)
- CSF penetration: low
- Adverse effects: hypersensitivity, interstitial nephritis, hypokalemia, phlebitis
Ampicillin
- Usual Adult Dose: 2 g every 4–6 hours IV
- Renal/hepatic failure: decrease dose in renal failure
- CSF penetration: low (13–14% with inflamed meninges)
- Adverse effects: hypersensitivity, maculopapular rash (especially with EBV infection)
Ampicillin-Sulbactam
- Usual Adult Dose: 1.5–3 g every 6 hours IV
- Renal/hepatic failure: decrease dose in renal failure
- CSF penetration: low
- Adverse effects: hypersensitivity, diarrhea
Amoxicillin
- Usual Adult Dose: 500 mg to 1 g every 8–12 hours orally
- Renal/hepatic failure: decrease dose in renal failure (500 mg q12h when CrCl 10–29 mL/min)
- CSF penetration: low
- Adverse effects: hypersensitivity
Amoxicillin-Clavulanate
- Usual Adult Dose: 500 mg every 8 hours or 875 mg every 12 hours orally
- Renal/hepatic failure: decrease dose in renal failure; avoid in severe hepatic impairment
- CSF penetration: low
- Adverse effects: hypersensitivity, diarrhea, hepatotoxicity (mainly clavulanate)
Piperacillin-Tazobactam
- Usual Adult Dose: 3.375–4.5 g every 6–8 hours IV; consider extended infusion (4.5 g over 4 hours q8h)
- Renal/hepatic failure: decrease dose in renal failure (3 g q12h when CrCl 10–29 mL/min)
- CSF penetration: low
- Adverse effects: hypersensitivity, platelet dysfunction, hypokalemia
Introduction: History and Impact of Penicillins
Penicillin was discovered by Alexander Fleming in September 1928 at St. Mary’s Hospital, London. Observing bacterial lysis around a contaminating Penicillium mold on a staphylococcal culture plate, he named the antibacterial substance “penicillin” and published his findings in 1929 (3). Unable to purify the active compound himself, clinical use had to wait until 1940, when Howard Florey and Ernst Chain at Oxford developed the purification process (2). The first human treated was a policeman with staphylococcal sepsis in 1941. Mass production—driven by the demands of World War II—began in 1943, and Fleming, Florey, and Chain received the Nobel Prize in 1945.
| Year | Milestone |
|---|---|
| 1928 | Fleming discovers penicillin |
| 1929 | Fleming publishes findings |
| 1940 | Florey & Chain purify penicillin |
| 1941 | First human treated |
| 1943 | Mass production begins |
| 1945 | Nobel Prize awarded |
The clinical impact was transformative. Before penicillin, bacterial endocarditis was virtually 100% fatal, pneumococcal pneumonia carried roughly 30% mortality, and staphylococcal sepsis was often a death sentence. Despite being nearly a century old, penicillins—along with their semisynthetic derivatives and β-lactamase inhibitor combinations—remain essential antibiotics today.
Chemistry
Basic Penicillin Structure
The penicillin nucleus consists of three structural components (Figure 1):
- Thiazolidine ring — a 5-membered ring containing sulfur that provides structural stability
- β-Lactam ring — a 4-membered ring under considerable geometric strain; essential for antibacterial activity and the target of β-lactamases
- Side chain (R group) — a variable component attached to the β-lactam nitrogen that determines the antibacterial spectrum, acid stability, β-lactamase stability, protein binding, and pharmacokinetic properties of each individual agent
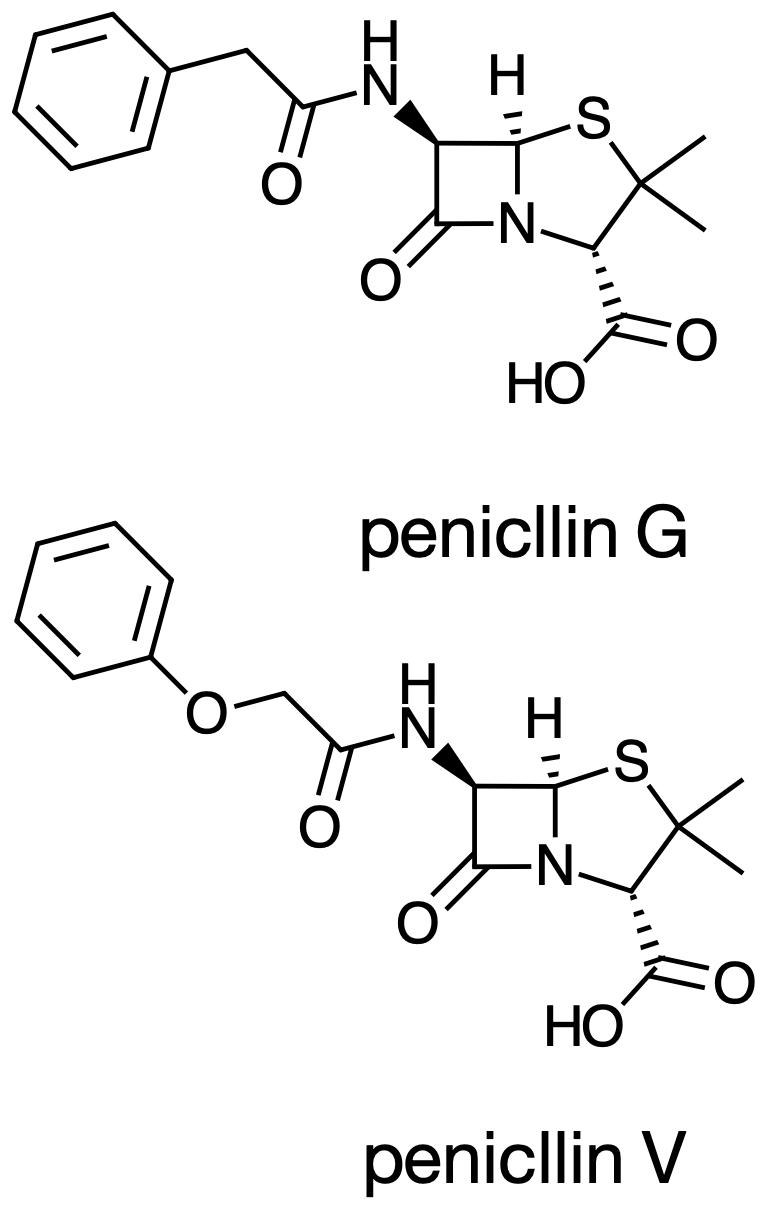
The β-Lactam Ring: Why It Matters
The β-lactam ring is the pharmacophore of all penicillins. Its four-membered geometry creates molecular strain, making it chemically reactive. Structurally, the β-lactam ring mimics the D-alanyl-D-alanine (D-Ala-D-Ala) terminus of the peptidoglycan pentapeptide—the natural substrate of penicillin-binding proteins (PBPs). When the β-lactam ring opens—whether by covalent binding to a PBP or by hydrolysis from a β-lactamase—antibacterial activity is lost.
No intact β-lactam ring = No antibacterial activity.
The Side Chain: Determining Properties
Modifications to the side chain explain nearly all differences among penicillin classes:
- Acid stability — determines whether oral absorption is feasible (e.g., penicillin V is acid-stable; penicillin G is not)
- β-Lactamase stability — a bulky side chain near the β-lactam ring provides steric hindrance against enzymatic attack (e.g., oxacillin, nafcillin)
- Spectrum of activity — an amino group improves penetration through gram-negative outer membranes (aminopenicillins)
- Protein binding — varies from ~17% (ampicillin) to ~97% (dicloxacillin); only free drug is pharmacologically active
- Cross-reactivity — side chain similarity, more than the β-lactam core itself, determines the risk of allergic cross-reactions with other β-lactams
6-Aminopenicillanic Acid (6-APA): The Semisynthetic Era
The isolation of 6-aminopenicillanic acid (6-APA)—the penicillin nucleus without a side chain—from Penicillium chrysogenum fermentation was a breakthrough that enabled creation of all modern semisynthetic penicillins. By attaching different side chains to 6-APA, chemists produced methicillin (β-lactamase–resistant), ampicillin (expanded gram-negative spectrum), and carbenicillin (antipseudomonal activity), among others.
Mechanism of Action
Overview
Penicillins are bactericidal antibiotics that target the final step of bacterial peptidoglycan synthesis. They kill only actively growing bacteria that are synthesizing new cell wall; dormant or slow-growing cells (persisters, biofilm-embedded organisms) can survive exposure.
Why Bacteria Need Cell Walls
Bacteria maintain high internal osmotic pressure. The cell wall—composed of peptidoglycan—counteracts this pressure and prevents osmotic lysis. Without it, bacteria swell and rupture. Human cells lack peptidoglycan entirely, which is why β-lactams have an excellent selective toxicity profile.
- Gram-positive bacteria: thick peptidoglycan layer (50–100 molecules)
- Gram-negative bacteria: thin peptidoglycan layer (1–2 molecules) plus an outer membrane lipopolysaccharide barrier
Peptidoglycan Structure and Transpeptidation
The structural subunit of peptidoglycan is a disaccharide of N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM), with a pentapeptide stem attached to NAM that terminates in D-Ala-D-Ala. Cross-links formed between adjacent pentapeptide stems—by the transpeptidation reaction—give the cell wall its mechanical strength.
During transpeptidation (Figure 2):
- Transpeptidase (PBP) binds the D-Ala-D-Ala terminus of the donor peptide
- A covalent acyl-enzyme intermediate forms with the penultimate D-Ala
- The terminal D-Ala is released
- Cross-link forms with the acceptor chain
Penicillin structurally mimics D-Ala-D-Ala and is accommodated by the PBP active site, but the resulting acyl-enzyme complex undergoes only very slow deacylation—leaving the enzyme irreversibly inhibited.
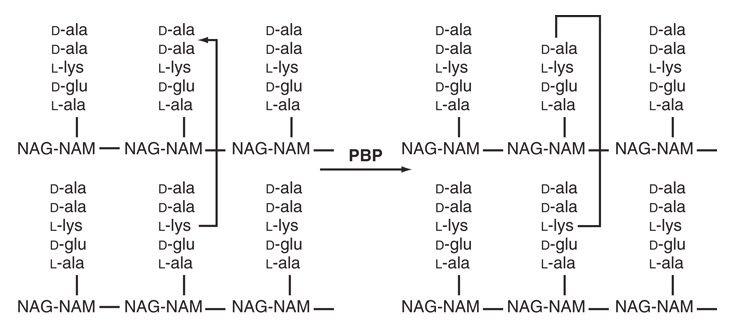
Penicillin-Binding Proteins (PBPs)
PBPs are membrane-bound serine enzymes found in all bacteria. They are evolutionarily related to β-lactamases; the critical difference is the rate at which the acyl-enzyme intermediate is hydrolyzed:
- PBPs: Slow deacylation → enzyme remains inhibited → cell wall synthesis fails
- β-Lactamases: Rapid deacylation → enzyme regenerates immediately → drug is destroyed (resistance)
| PBP Class | Size | Function |
|---|---|---|
| High-MW Class A | >50 kDa | Bifunctional: transglycosylase + transpeptidase |
| High-MW Class B | >50 kDa | Transpeptidase only |
| Low-MW | <50 kDa | Carboxypeptidases |
| PBP (in E. coli) | Function | Effect of Inhibition |
|---|---|---|
| PBP1a/1b | Transglycosylase + transpeptidase | Rapid cell lysis |
| PBP2 | Cell elongation, rod shape | Round cells |
| PBP3 | Septum formation, cell division | Long filaments |
| PBP4–6 | Carboxypeptidases | Minor effects |
Different β-lactams have different affinities for these PBPs, explaining why some agents cause rapid lysis while others induce filamentation.
β-Lactamases are evolutionarily derived from PBPs. The key biochemical distinction is not binding affinity but deacylation rate: PBPs hold on; β-lactamases release drug rapidly and regenerate.
Tolerance vs. Resistance
It is clinically important to distinguish resistance from tolerance:
- Resistance: Bacteria grow in the presence of the antibiotic (MIC is elevated)
- Tolerance: Bacteria survive but do not grow despite a low MIC; the minimum bactericidal concentration (MBC) is disproportionately high compared to the MIC. Stationary-phase cells and biofilm-embedded persister cells are classic examples.
Tolerance explains why some infections—particularly endocarditis and biofilm-associated infections—can relapse despite an organism that appears “susceptible” on routine susceptibility testing.
Resistance Mechanisms
Four mechanisms account for clinically significant resistance to penicillins and other β-lactams:
- β-Lactamase production — enzymatic destruction of the β-lactam ring (most common)
- Decreased outer membrane permeability — porin mutations in gram-negatives
- Active efflux — energy-dependent drug removal across the outer membrane
- Altered PBPs — reduced affinity for β-lactam binding
Multiple resistance mechanisms frequently coexist—especially in multidrug-resistant gram-negatives such as Pseudomonas aeruginosa—making individual mechanisms harder to predict clinically.
Mechanism 1: β-Lactamases
β-Lactamases are the most common and clinically important resistance mechanism. These enzymes hydrolyze the β-lactam ring, converting the active antibiotic to an inactive penicilloic acid derivative. The catalytic cycle (acylation → rapid hydrolysis → enzyme regeneration) means that a single β-lactamase molecule can destroy thousands of antibiotic molecules.
In gram-positive bacteria, β-lactamases are secreted extracellularly. In gram-negative bacteria, they are sequestered in the periplasmic space, where they intercept drug before it can reach PBPs.
Ambler Classification
β-Lactamases are classified into four molecular classes (A–D) based on amino acid sequence homology (Table 4):
| Ambler Class | Active Site | Mechanism | Key Examples | Inhibitors |
|---|---|---|---|---|
| A | Serine | Acyl-enzyme intermediate | TEM-1, SHV-1, CTX-M (ESBLs), KPC (carbapenemase) | Clavulanic acid, avibactam, vaborbactam, relebactam |
| B | Zinc (metallo) | Direct hydrolysis (Zn²⁺) | NDM-1, VIM-2, IMP-1 | None currently approved |
| C | Serine | Acyl-enzyme intermediate | AmpC, CMY-2 | Avibactam, relebactam, vaborbactam |
| D | Serine | Acyl-enzyme intermediate | OXA-48, OXA-23 | Avibactam (OXA-48 only) |
Class A: The Most Common Enzymes
Class A enzymes include the most widespread β-lactamases:
- TEM-1: Most common plasmid-encoded β-lactamase worldwide; hydrolyzes penicillins and narrow-spectrum cephalosporins; inhibited by clavulanic acid
- SHV-1: Common in Klebsiella; similar spectrum to TEM-1
- CTX-M: Now the dominant extended-spectrum β-lactamase (ESBL) globally; point mutations expanded substrate range to include extended-spectrum cephalosporins and aztreonam
- KPC: A serine carbapenemase; hydrolyzes carbapenems; inhibited by avibactam, vaborbactam, and relebactam (but not classic inhibitors clavulanate or tazobactam reliably)
Extended-Spectrum β-Lactamases (ESBLs)
ESBLs are Class A enzymes that have acquired the ability to hydrolyze extended-spectrum cephalosporins (cefotaxime, ceftriaxone, ceftazidime) and aztreonam. They are plasmid-encoded and therefore spread easily. Although ESBLs are inhibited by clavulanic acid in vitro, clinical outcomes with β-lactam/β-lactamase inhibitor combinations for serious ESBL infections (e.g., bacteremia) can be unpredictable due to the inoculum effect and pharmacodynamic limitations.
Class B: Metallo-β-Lactamases (MBLs)
MBLs use zinc ions rather than an active-site serine. They hydrolyze virtually all β-lactams except aztreonam (monobactam). No currently approved β-lactamase inhibitor is active against MBLs. Major MBLs include NDM-1 (New Delhi metallo-β-lactamase, now distributed globally), VIM (common in Europe and P. aeruginosa), and IMP. The inability of MBLs to hydrolyze aztreonam is being exploited clinically with aztreonam–avibactam combinations.
Class B metallo-β-lactamases cannot hydrolyze aztreonam (monobactam). This is being exploited in novel combination regimens (aztreonam + avibactam) for NDM-producing organisms.
Class C: AmpC β-Lactamases
AmpC-type enzymes preferentially hydrolyze cephalosporins and are often chromosomally encoded and inducible. Exposure to certain β-lactams (e.g., ceftazidime, cefoxitin, imipenem) can induce high-level AmpC expression, leading to clinical failure despite initial susceptibility.
The classic “SPACE” mnemonic (Serratia, Pseudomonas, Acinetobacter, Citrobacter, Enterobacter) describes organisms historically associated with inducible chromosomal AmpC, but this mnemonic is imprecise and may lead to overtreatment. A more clinically relevant mnemonic is “HECK-YES”:
Hafnia, Enterobacter, Citrobacter (freundii complex), Klebsiella aerogenes, Yersinia, Enterobacter, Serratia
These organisms carry clinically meaningful inducible chromosomal AmpC. Traditional β-lactamase inhibitors (clavulanate, tazobactam) do not inhibit AmpC; avibactam, relebactam, and vaborbactam do.
Class D: OXA Enzymes
OXA enzymes are named for their ability to hydrolyze oxacillin. They are a heterogeneous group:
- OXA-48: An important carbapenemase in Klebsiella pneumoniae, predominantly in the Middle East and Europe; inhibited by avibactam but not by traditional inhibitors
- OXA-23 / OXA-24: Major carbapenemases in Acinetobacter baumannii
OXA-48 is particularly challenging because it only modestly elevates carbapenem MICs, which can delay recognition.
Carbapenemases: Summary
| Enzyme | Ambler Class | Geographic Distribution | Inhibitors |
|---|---|---|---|
| KPC | A | Americas, worldwide | Avibactam, vaborbactam, relebactam |
| NDM | B | South Asia, global | None approved |
| VIM | B | Europe, global | None approved |
| OXA-48 | D | Middle East, Europe | Avibactam |
Mechanism 2: Decreased Outer Membrane Permeability
This mechanism is relevant only for gram-negative bacteria. Hydrophilic β-lactams must traverse the outer membrane through protein channels called porins. Mutations that reduce porin expression or alter channel size reduce intracellular drug accumulation. In Pseudomonas aeruginosa, loss of the OprD porin is a classic mechanism of carbapenem (particularly imipenem) resistance. Permeability loss alone usually causes only modest, low-level resistance but is clinically important when combined with β-lactamase production or efflux.
Mechanism 3: Efflux Pumps
Efflux pumps actively transport β-lactams (and other antibiotics) out of the bacterial cell, using energy from the proton motive force or ATP hydrolysis. They often have broad substrate specificity, contributing to multidrug resistance. The MexAB-OprM system in P. aeruginosa is a prototypical example. Efflux is particularly significant when combined with β-lactamase production and reduced permeability.
Mechanism 4: Altered PBPs
PBPs with reduced affinity for β-lactams allow cell wall synthesis to continue despite antibiotic exposure. Key examples:
- MRSA: Carries the mecA gene (on the mobile SCCmec element), which encodes PBP2a—a novel PBP with very low affinity for nearly all β-lactams. Because PBP2a is not inhibited, cell wall synthesis continues even when native PBPs are saturated. Note: MRSA resistance is not due to β-lactamase production; adding a β-lactamase inhibitor will not restore activity. The only β-lactams with clinically useful activity against MRSA are ceftaroline and ceftobiprole, which have enhanced binding to PBP2a.
- Penicillin-resistant S. pneumoniae: Mosaic PBP genes acquired through transformation produce PBPs with reduced affinity
- Enterococcus faecium: Intrinsic low-affinity PBP5 explains its resistance to most β-lactams (but E. faecalis retains PBPs susceptible to ampicillin)
MRSA resistance to β-lactams is mediated by PBP2a (encoded by mecA), not by β-lactamase production. β-Lactamase inhibitors are ineffective against MRSA.
Classification of Penicillins
Penicillins are divided into five classes based on antibacterial spectrum:
- Natural penicillins — penicillin G, penicillin V
- Penicillinase-resistant penicillins — nafcillin, oxacillin, dicloxacillin, flucloxacillin
- Aminopenicillins — ampicillin, amoxicillin
- Carboxypenicillins — ticarcillin (largely obsolete)
- Acylureidopenicillins — piperacillin
Class 1: Natural Penicillins
Penicillin G (parenteral) and penicillin V (oral) are the original, narrowest-spectrum penicillins—but they have the highest potency against susceptible organisms. Penicillin G is acid-labile (unsuitable for oral use); penicillin V has an acid-stable side chain. Both are susceptible to β-lactamases.
Spectrum of activity:
- Streptococcus pyogenes (Group A strep) — essentially 100% susceptibility maintained
- Streptococcus agalactiae (Group B strep)
- Streptococcus pneumoniae (susceptible strains)
- Neisseria meningitidis
- Treponema pallidum (syphilis)
- Oral anaerobes
- Listeria monocytogenes
| Infection | Preferred Agent |
|---|---|
| Group A strep pharyngitis | Penicillin V (oral) |
| Syphilis (all stages except neuro) | Benzathine penicillin G IM |
| Neurosyphilis | High-dose IV penicillin G |
| Meningococcal meningitis | IV penicillin G (or ceftriaxone empirically) |
| Actinomycosis | IV penicillin G |
| Gas gangrene (C. perfringens) | IV penicillin G |
Penicillin G remains the drug of choice for syphilis at all stages, including in pregnancy. Penicillin-allergic pregnant patients should undergo desensitization; doxycycline and azithromycin are not acceptable alternatives for neurosyphilis.
Class 2: Penicillinase-Resistant Penicillins (Antistaphylococcal Penicillins)
Bulky side chains create steric hindrance that physically blocks β-lactamase access to the β-lactam ring. These agents are active against MSSA and streptococci but have no meaningful gram-negative coverage. Critically, they are not active against MRSA (whose resistance is PBP2a–mediated, not β-lactamase–mediated).
| Drug | Route | Protein Binding | Elimination | Special Considerations |
|---|---|---|---|---|
| Nafcillin | IV | ~90% | Primarily hepatic | No renal dose adjustment; hypokalemia, phlebitis |
| Oxacillin | IV | ~90% | Mixed hepatic/renal | Hepatotoxicity; interstitial nephritis |
| Dicloxacillin | PO | ~96% | Mixed | Highest protein binding; take on empty stomach |
| Flucloxacillin | PO/IV | ~96% | Primarily renal | Not available in USA; widely used in Europe |
Nafcillin is the only penicillin that does not require dose adjustment in renal failure.
Class 3: Aminopenicillins
The addition of an amino group to the side chain improves penetration through gram-negative outer membrane porins, expanding spectrum compared to natural penicillins. Both agents are susceptible to β-lactamases.
Expanded spectrum relative to natural penicillins:
- Enterococcus faecalis
- Haemophilus influenzae (non–β-lactamase–producing strains)
- Escherichia coli (non–β-lactamase–producing strains)
- Proteus mirabilis
- Salmonella and Shigella spp.
- Listeria monocytogenes
Ampicillin vs. Amoxicillin:
| Property | Ampicillin | Amoxicillin |
|---|---|---|
| Oral bioavailability | 30–55% | 74–92% |
| Effect of food | Decreased | None |
| Preferred route | IV | Oral |
For oral therapy, amoxicillin is almost always preferred over ampicillin. Note that at higher doses (≥1 g), amoxicillin’s fractional absorption decreases (~60–70%), though total drug exposure still increases.
Classes 4 & 5: Antipseudomonal Penicillins
Carboxypenicillins (ticarcillin) extended gram-negative and antipseudomonal coverage but are largely obsolete. Piperacillin (acylureidopenicillin) has broader gram-negative and excellent anaerobic activity and is now used exclusively combined with the β-lactamase inhibitor tazobactam.
Spectrum Summary
| Class | Gram-positive | Gram-negative | Pseudomonas | Anaerobes | MRSA |
|---|---|---|---|---|---|
| Natural penicillins | +++ | − | − | ++ | − |
| Antistaphylococcal | ++ (MSSA) | − | − | − | − |
| Aminopenicillins | ++ | + | − | ++ | − |
| Antipseudomonal | + | ++ | ++ | +++ | − |
None of the penicillins are active against MRSA. Only ceftaroline and ceftobiprole (anti-MRSA cephalosporins) have useful β-lactam activity against MRSA.
Pharmacologic Properties
Oral Absorption
| Penicillin | Absorption (%) | Food Effect |
|---|---|---|
| Penicillin G | Negligible | Acid-labile |
| Penicillin V | ~60% | None |
| Ampicillin | 30–55% | Decreased |
| Amoxicillin | 74–92% | None |
| Dicloxacillin | ~37% | Decreased |
| Flucloxacillin | ~44% | Decreased |
Protein Binding
Protein binding ranges from approximately 17% (aminopenicillins) to 97% (dicloxacillin). Only free (unbound) drug is pharmacologically active and able to reach tissues. High protein binding prolongs half-life but may reduce free drug concentrations at sites of infection. For penicillins with >90% protein binding, this can have clinical implications, particularly for deep-seated infections.
Distribution and CNS Penetration
Penicillins generally achieve therapeutic concentrations in lungs, liver, kidneys, muscle, and pleural, peritoneal, and synovial fluids. CNS penetration under normal conditions is poor for all penicillins; inflammation of the meninges increases penetration significantly, though still limited:
| Condition | Penicillin G | Ampicillin |
|---|---|---|
| Normal meninges | <1% | <1% |
| Inflamed meninges | 5–10% | 13–14% |
Because CNS penetration improves with meningeal inflammation, high-dose IV therapy is required for meningitis. As inflammation resolves with successful treatment, drug penetration also decreases—providing a pharmacologic rationale for maintaining treatment duration even as patients improve clinically.
Elimination and Renal Dosing
Most penicillins are excreted by the kidneys via glomerular filtration and active tubular secretion, with short half-lives (0.5–1.5 hours). Probenecid can block tubular secretion, prolonging drug exposure. Nafcillin (primarily hepatic) and partially oxacillin are exceptions and do not require renal dose adjustment.
| Agent | CrCl 10–29 mL/min | Hemodialysis |
|---|---|---|
| Penicillin G | 75% of usual dose | Dose after HD |
| Ampicillin | 0.5–2 g q12h | 0.5–1 g q12–24h |
| Amoxicillin | 500 mg q12h | 500 mg q12–24h |
| Piperacillin-tazobactam | 3.375 g q8–12h | 3.375 g q12h; dose after HD |
| Nafcillin | No change | No change |
PK/PD Optimization: Time-Dependent Killing
β-Lactams are time-dependent antibiotics: killing efficacy correlates with the fraction of the dosing interval that free drug concentrations remain above the MIC (%T>MIC). The PK/PD target for bactericidal activity is typically 40–70% T>MIC. Strategies to optimize this include:
- More frequent dosing (e.g., q4h rather than q6h)
- Extended infusions: Infusing the dose over 3–4 hours (rather than 30 minutes) maintains concentrations above the MIC for a greater fraction of the dosing interval
| Regimen | Infusion Duration | Peak | %T>MIC (est.) |
|---|---|---|---|
| Traditional (pip-tazo 4.5 g q6h) | 30 min | High | Lower |
| Extended infusion (pip-tazo 4.5 g q8h) | 4 hours | Lower | Higher |
Extended infusion piperacillin-tazobactam (e.g., 4.5 g over 4 hours q8h) is now standard of care in many institutions, particularly for serious infections caused by organisms with elevated MICs (e.g., Pseudomonas aeruginosa).
Adverse Effects
Hypersensitivity Reactions
Hypersensitivity is the most clinically significant class of adverse effects. Penicillins act as haptens by binding covalently to proteins, triggering immune responses.
| Type | Timing | Mechanism | Manifestations |
|---|---|---|---|
| Type I (IgE-mediated) | Minutes–hours | IgE, mast cell degranulation | Anaphylaxis, urticaria, angioedema, bronchospasm |
| Type II (cytotoxic) | Days | Antibody-mediated | Hemolytic anemia, cytopenias |
| Type III (immune complex) | 1–3 weeks | Immune complex deposition | Serum sickness, drug fever |
| Type IV (delayed T-cell) | Days–weeks | T-cell–mediated | Maculopapular rash, contact dermatitis |
Type I reactions are the most dangerous. Importantly, Type IV delayed maculopapular rashes (the most common reported reaction) generally do not preclude future use and are frequently mislabeled as “penicillin allergy.”
Penicillin Allergy: The Numbers
- ~10% of patients report a penicillin allergy
- <1% have confirmed IgE-mediated allergy when formally tested
- ~2% react when challenged
- True anaphylaxis: <0.01%
- Sensitization wanes over time: ~50% lose reactivity within 5 years; ~80% within 10 years
The vast majority of “penicillin allergy” labels are inaccurate. Over-labeling leads to use of broader-spectrum, more toxic, and more expensive alternatives, with worse clinical outcomes.
Penicillin Allergy De-Labeling
De-labeling is a key antimicrobial stewardship intervention:
- History assessment: Determine whether the reaction had features of true allergy (timing, symptoms, severity)
- Risk stratification: High-risk vs. low-risk features
- Skin testing: Identifies IgE-mediated sensitization
- Graded oral challenge: Confirms tolerance in negative-testing or low-risk patients
- Record update: Correct the allergy label in the medical record
Penicillin allergy de-labeling programs are safe and significantly improve patient outcomes by restoring access to preferred, narrower-spectrum agents.
Cross-Reactivity with Other β-Lactams
Historical estimates of 10% cross-reactivity between penicillins and cephalosporins were based on flawed data (penicillin contamination of early cephalosporin preparations). Current evidence suggests the rate is ~1–2%. Cross-reactivity is driven more by R1 side chain similarity than by the shared β-lactam ring:
- Higher risk: Ampicillin/amoxicillin → cephalexin or cefadroxil (similar side chains)
- Lower risk: Ampicillin/amoxicillin → ceftriaxone or cefepime (dissimilar side chains)
Carbapenems and aztreonam have very low cross-reactivity with penicillins; aztreonam shares a side chain with ceftazidime, creating a clinically relevant cross-reactivity pair.
Agent-Specific Adverse Effects
| Effect | Most Common With | Notes |
|---|---|---|
| Maculopapular rash | Ampicillin | Near-universal with infectious mononucleosis (EBV); not true allergy |
| Diarrhea | Ampicillin, amoxicillin-clavulanate | Gut flora disruption; clavulanate component implicated |
| Hepatotoxicity | Oxacillin, amoxicillin-clavulanate | Clavulanate-associated hepatotoxicity can be delayed; check LFTs |
| Neutropenia | Prolonged high-dose therapy | Reversible; monitor CBC with courses >2 weeks |
| Seizures | High-dose penicillin G | Risk increased in renal failure due to accumulation |
| Interstitial nephritis | Nafcillin, oxacillin (methicillin) | Immune-mediated; monitor urinalysis |
| Hypokalemia | Nafcillin, piperacillin | Cation exchange effect |
| Hyperkalemia | Penicillin G potassium salt | At very high doses |
| Platelet dysfunction | Piperacillin | With prolonged courses; monitor in surgical patients |
Individual Penicillins
Aminopenicillins
The antibacterial activities of aminopenicillins are similar (Figure 3). They are susceptible to hydrolysis by β-lactamases.
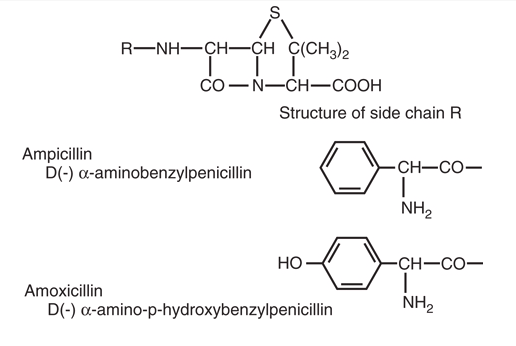
Ampicillin
Ampicillin is available as 250- or 500-mg capsules for oral use and in parenteral formulations. For most indications where oral therapy is appropriate, amoxicillin is preferred because of greater bioavailability. IV ampicillin remains important for enterococcal infections, Listeria meningitis, and empiric therapy for neonatal sepsis.
Amoxicillin
Amoxicillin differs from ampicillin only in the addition of a hydroxyl group in the para position of the benzene side chain, which significantly improves intestinal absorption (74–92% vs. 33–54%). Amoxicillin can be taken with food without reduced absorption.
High-dose amoxicillin (80–90 mg/kg/day in divided doses) is first-line therapy for otitis media in children, as these doses overcome the MIC for penicillin-intermediate and penicillin-resistant S. pneumoniae at the site of infection.
Antipseudomonal Penicillins
Piperacillin
Piperacillin has broad gram-negative coverage including P. aeruginosa (Figure 4), excellent anaerobic coverage, and gram-positive activity comparable to aminopenicillins. In current practice, piperacillin is used exclusively in the combination formulation piperacillin-tazobactam to extend activity against β-lactamase–producing strains. Extended-infusion dosing is widely recommended for serious infections.
β-Lactam and β-Lactamase Inhibitor Combinations
Classes of β-Lactamase Inhibitors
Six β-lactamase inhibitors are currently in clinical use, divided into two generations:
Traditional (β-lactam-based) inhibitors — act as “suicide substrates” that irreversibly acylate and inactivate β-lactamases; one inhibitor molecule inactivates one enzyme molecule:
- Clavulanic acid (with amoxicillin or ticarcillin)
- Sulbactam (with ampicillin); note sulbactam has intrinsic activity against Acinetobacter baumannii
- Tazobactam (with piperacillin)
Novel (non-β-lactam) inhibitors — structurally distinct, with broader coverage including AmpC and some carbapenemases:
- Avibactam (with ceftazidime)
- Relebactam (with imipenem-cilastatin)
- Vaborbactam (with meropenem)
Spectrum of Inhibition
| Inhibitor | Class A (TEM/SHV) | ESBLs | KPC | AmpC (Class C) | MBLs (Class B) | OXA-48 (Class D) |
|---|---|---|---|---|---|---|
| Clavulanate | ✓ | ± | ✗ | ✗ | ✗ | ✗ |
| Sulbactam | ✓ | ± | ✗ | ✗ | ✗ | ✗ |
| Tazobactam | ✓ | ± | ✗ | ✗ | ✗ | ✗ |
| Avibactam | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ |
| Relebactam | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ |
| Vaborbactam | ✓ | ✓ | ✓ | ✓ | ✗ | ✗ |
No approved β-lactamase inhibitor is active against Class B metallo-β-lactamases (NDM, VIM, IMP). Aztreonam–avibactam is under investigation for this clinical gap.
Clinical Limitations of β-Lactam/β-Lactamase Inhibitor Combinations
In vitro susceptibility does not guarantee clinical success. Key limitations include:
- ESBL infections: Treatment failures have been reported with traditional β-lactam/β-lactamase inhibitor combinations (amoxicillin-clavulanate, piperacillin-tazobactam) for serious ESBL infections such as bacteremia. Carbapenems are generally preferred for ESBL bacteremia.
- Inoculum effect: At high bacterial densities, β-lactamase enzyme levels may exceed inhibitor concentrations, overwhelming inhibition and restoring resistance.
- AmpC producers: Traditional inhibitors are ineffective against AmpC (Class C); pip-tazo may appear active in vitro but is unreliable for serious infections with AmpC-inducible organisms.
- Carbapenemase producers: Organism-specific inhibitor selection is critical; no single inhibitor covers all carbapenemases.
Clinical Application
Quick Selection Guide
| Infection | First-Line Penicillin (or Combination) |
|---|---|
| Strep pharyngitis | Penicillin V |
| MSSA skin/soft tissue infection | Dicloxacillin (oral) or nafcillin/oxacillin (IV) |
| MSSA bacteremia/endocarditis | Nafcillin 2 g q4h IV or cefazolin 2 g q8h IV |
| Syphilis (primary/secondary/latent) | Benzathine penicillin G 2.4 MU IM × 1 |
| Neurosyphilis | Penicillin G 18–24 MU/day IV × 10–14 days |
| Meningococcal meningitis | Ceftriaxone empirically; IV penicillin G if confirmed susceptible |
| Listeria meningitis | Ampicillin 2 g q4h IV |
| CAP (outpatient, no comorbidities) | Amoxicillin 1 g q8h |
| CAP (outpatient, COPD or comorbidities) | Amoxicillin-clavulanate 875 mg q12h |
| Community-acquired intra-abdominal infection | Piperacillin-tazobactam 4.5 g q6–8h IV (extended infusion) |
| Uncomplicated cystitis | Nitrofurantoin or TMP-SMX preferred (ampicillin resistance >40% in E. coli) |
MSSA Endocarditis: Nafcillin vs. Cefazolin
An important clinical nuance is the comparison of antistaphylococcal penicillins versus cefazolin for MSSA bacteremia and endocarditis. Multiple observational studies and a recent randomized trial (CloCeBa) suggest cefazolin is noninferior and may offer advantages including lower nephrotoxicity, fewer drug fever events, and simpler dosing (1,4,5). Many institutions now consider cefazolin a preferred or equivalent alternative to nafcillin/oxacillin for MSSA infections.
Vancomycin is significantly inferior to both antistaphylococcal penicillins and cefazolin for MSSA bacteremia and endocarditis. Reserve vancomycin for patients with confirmed penicillin allergy (IgE-mediated) or MRSA.
Summary
Key Takeaways
Structure determines function: The β-lactam ring is essential for activity; side chain modifications create the five penicillin classes with distinct spectrum, stability, and PK profiles.
Mechanism: Penicillins inhibit transpeptidation by covalently binding PBPs and preventing peptidoglycan cross-linking. They are bactericidal only against actively growing cells.
Four resistance mechanisms: β-Lactamase production (most common), decreased outer membrane permeability, efflux pumps, and altered PBPs (MRSA = PBP2a, not β-lactamase).
β-Lactamases matter: Know the Ambler classes and their inhibitor susceptibilities. Traditional inhibitors (clavulanate, tazobactam) cover Class A but not AmpC, MBLs, or KPC reliably. Newer inhibitors (avibactam, vaborbactam, relebactam) cover KPC and AmpC but not MBLs.
Match spectrum to pathogen: Use the narrowest effective penicillin. No penicillin covers MRSA. Aminoglycosides and β-lactam combinations may be needed for difficult gram-negatives.
PK/PD optimization: β-Lactams are time-dependent killers (%T>MIC); extended infusions improve pharmacodynamics for organisms with higher MICs.
De-label penicillin allergies: Most labeled allergies (~10% of patients) are not true IgE-mediated allergies. Skin testing and oral challenges can safely de-label most patients.